SPACE· STUDIES INSTITUTE
P.Q BOX 82
PRINCETON, NEW JERSEY 08540
[[librarian note: This address is here, as it was in the original printed newsletter, for historical reasons. It is no longer the physical address of SSI. For contributions, please see this page]]
SSI UPDATE
THE HIGH FRONTIER® NEWSLETTER
VOLUME XII ISSUE 5 SEPTEMBER/OCTOBER 1986
VICE PRESIDENT’S COLUMN
As we go to press, we have just learned of the report entitled Planetary Exploration to the Year 2000, An Augmented Program. It is a second volume in a two-volume report by the Solar System Exploration Committee of the NASA Advisory Council. The first volume of the report was entitled A Core Program, and it serves as the basis for most of the current thinking about the future of U.S. planetary exploration.
As its name suggests, this second volume discusses advanced space activity beyond the bare bones program of planetary research. From our standpoint, its most exciting projections occur in Chapter 6, entitled The Riches of Space. This chapter lays out a concise summary of our present knowledge of nonterrestrial resources and how they might be used in future space projects, including solar power satellites, rocket propellant, and shielding. Both lunar and asteroidal resources are described as are the advantages and disadvantages of both of these categories of resources. The chapter contains an excellent synopsis of the main unanswered questions about the composition and use of non-terrestrial materials, and includes recommendations on the search for wild card resources, such as volatiles trapped at the Moon’s poles (which is the subject of our Lunar Polar Probe study, to be reported in a future SSI Update).
Another excellent feature of this new book is its inclusion of near term recommendations. One of the foremost of these is that it is suggested that the Space Station be planned and established in ways that can support the use of non-terrestrial materials. The chapter includes discussion on mining helium-3 on the Moon. The entire volume is beautifully illustrated with the Chapter on non-terrestrial resources being no exception. One particular favorite is a William Hartman painting of a mass-driver reaction engine delivering a chunk of asteroid to near-earth space. The report is now available from the Government Printing Office book store (see information on page 4).
It is especially heartening to see the work that all of us at SSI have supported becoming accepted and widely discussed in forms such as these. The fact that this report and reports like the National Commission on Space study can confidently discuss mass-drivers and chemical processing of lunar soil is due to your efforts. Let’s keep pulling together to reduce the number of unanswered questions and accelerate the human breakout into space.
Gregg Maryniak
A Special Thank You
On Saturday, August 2nd, a dedicated group of SSI supporters spent the day moving the SSI office equipment, library and MassDriver to our new quarters in Rocky Hill. We would like to extend a special thank you to Senior Associates: Morris Hornik, Rick Tumlinson, Bob Summersgill, Dennis Mateik, Randy Gigante and Ray Hoover. Thanks also to Howard Ziserman and Carole Kingsbury.
RESEARCH REPORT
COMPOSITES: Fibers and Matrices from Lunar Regolith
By Charles H. Coggin, Jr.
President of Glass Strand, Inc.
(Continued from last issue)
MARE BASALTS AS FIBER RAW MATERIAL
The mare basalts are igneous rocks derived from the interior of the Moon as melts by well known igneous processes. The mare basalts can be divided into two major chemical groups based on titanium content.
The general technology necessary to produce glass fibers from terrestrial basalts is well known. Earth basalts and lunar basalts are very similar and the proven processes used on Earth to fiberize basalts should work with lunar basalts. Basaltic rock on Earth contains compounds of iron, including substantial amounts of both ferrous oxide, (FeO) and ferric oxide, (Fe203). When the basaltic rocks are melted in air using an electric furnace and subsequently drawn through a platinum die, substantial portions of the ferrous oxide (FeO) is oxidized to ferric oxide (Fe203). It has been found that minimizing the ratio of ferric oxide to ferrous oxide in the resulting fibers improves the tensile strength. Therefore the properties of basalt fibers should be enhanced when produced on the Moon since no ferric oxide is present in lunar basalts and melting in a non-oxidizing environment requires no special apparatus.
Basaltic rock from Earth having an initial content of 2.1% ferric oxide and 11.5% ferrous oxide was melted under normal conditions and drawn to produce fibers of identical diameter. Fibers were being drawn in air and in a nitrogen atmosphere, which was utilized to prevent oxidation of the ferrous iron. Samples 1 and 2 were drawn in air, allowing oxidation of the ferrous iron without control. Sample 3 was drawn under a nitrogen atmosphere to prevent oxidation of the ferrous iron. The substantial improvements in tensile strength are readily observable.
Fibers produced from Earth basalts were found to be comparable to E-glass fibers in strength and modulus. The finest fibers have a silk-like sheen and are golden brown in color.
With increasing diameter the color darkens and fibers become brittle. Fibers produced from basalts do not have the consistent superior properties that can be achieved with the use of more refined materials. However, the properties are more than adequate for any structural need in the development of a lunar colony.
FELDSPAR AS FIBER RAW MATERIAL
A glass reinforcing fiber the equivalent of anything produced on Earth can be derived using feldspar as the principal raw material. Apollo missions to the lunar highlands have shown that feldspar is present in the regolith as well as in outcroppings. As export of composites becomes a primary interest, it will probably be more feasible to mine subsurface deposits rather than separate the feldspar float from surface regolith.
Feldspar is the most important source of alumina on the Moon. The mineral can be obtained with a reasonably constant composition and in a state of sufficient purity from iron and other objectionable elements. Feldspar usually melts between 1100-1200 C and melts readily in the process of glassmaking.
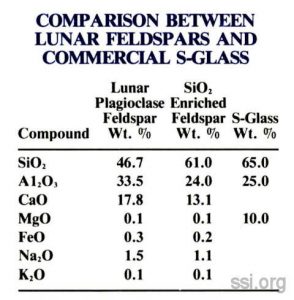
Feldspar is a complex aluminosilicate containing a mixture of CaO, Na20 and/or K2O. Lunar plagioclase feldspars are solid solutions of anorthite (CaAl2Si208) and albite (NaAlSi308).
If the native feldspar were enriched by an addition of 36% Si02, the resultant mixture would closely match the composition and properties of S-glass, the highest performance commercial glass fiber produced on Earth (see table on page 2). CaO and MgO can be substituted for each other without significantly altering the glass properties. Because of the other oxides present, the Si02 enriched feldspar would melt and fiberize better than the S-glass.
It may be seen that with presently available bushing materials and fiberizing techniques, glasses with good fiberizing characteristics can be obtained over a relatively small oval shaped composition region. The silica content in these glasses may vary between approximately 50% by weight and 70% by weight, the alumina between approximately 10% to 30% and the magnesia between approximately 10% and 25%.
Although the production of fibers for reinforcement from lunar raw materials appears to be relatively straightforward, the creation of matrix materials is considerably more challenging. On Earth, the matrix materials used in composites are normally organic with the exception of metal matrix composition. Since there are no organics on the lunar surface, the obvious task is to explore the possibility of an inorganic matrix material.
The matrix of a fibrous composite plays a key role both in developing and in maintaining the strength of the composite. The primary function of the matrix is to permit the reinforcement material to contribute its strength to the final structure. To perform this function, the matrix must first be able to wet the surface of the fiber. This requires a fairly high degree of fluidity at some period during the preparation of the composite.
Having obtained molecular contact, the matrix material must remain in contact with the fiber during curing, cooling, crystallization, or whatever other material change occurs during solidification. Finally, the matrix must remain strongly adhered to that surface after this solidification is completed. Because fluctuation temperature is usually associated with these curing and cooling processes, the thermal expansion of the matrix should be as close as possible to that of the fiber. Barring this, the matrix should be strong or flexible enough to redistribute the forces resulting from thermal change away from the interface. In composites loaded under pure tension, as in filamentwound pressure containers, such matching and redistribution are not as important because higher shrinkage of the matrix places the fiber under compression, which may act merely as a form of prestressing.
To perform the function of stress transfer, the matrix must have an elongation higher, and a modulus lower, than that of the fiber to permit the force to be effectively transferred to the fiber instead of being captured by the matrix itself.
The second important function of the matrix is to position and protect the load-carrying fibers. To accomplish this, the matrix must be highly resistant to any impressed environment, and also resist penetration of that environment to the sensitive interfacial area.
The third matrix function is to provide lateral support to the fibers for resistance to buckling under compressive loads. In addition the matrix must resist interlaminar shear fracture and transfer stresses from fiber to fiber and around discontinuities.
The development of glass fiber strengthened glasses in Earth laboratories has added a new dimension to composites. It is now possible to fabricate a wide range of high performance composite materials that combine the useful attributes of ceramics with the toughness of fiber reinforced composites.
The strength of glass composites are mostly determined by the strength of the glass filament. The fibers are the primary load-carrying element of the composite, because of their high modulus of elasticity relative to the modulus of the matrix and because of the relatively high volume of the fibers in the composite. The matrix acts to harden the shape, transfer loads from broken filaments, and protect the filaments from mechanical and chemical degradation.
Efficient use of the strength of fibrous composites requires that the fibers and matrix act together as a unit. This presumes that a good bond exists between the matrix and fibers. The bond need not be continuous, but the bonded points must be bonded together well enough to ensure that the two dissimilar materials can absorb strain as a unit. The key functions of the bond are to minimize or eliminate fiber slippage and to facilitate stress transfer through the matrix.
It is of particular importance in a glass/glass composite that both fiber and matrix be dimensionally stable. Most fibers have low coefficents of expansion during heating. As a consequence the resultant composites will exhibit very low coefficents of thermal expansion. These composites have the ability to retain their strength to temperatures well beyond current resin matrix systems.
Because glass can be treated as a thermoplastic material, many of the processes developed for fiber reinforced glasses can be made to emulate those previously used for polymer matrix systems. The ability to control the viscosity of matrix glasses and to deform them easily under pressure permits the physical densification of fiber reinforced composites without mechanical damage to the fibers. Also, the composite densification process can be rapid since glass matrix flow is all that is required. After full densification the glass matrix can be crystallized, if desired, under controlled conditions to achieve a matrix with superior toughness and high temperature strength.
Improved toughness after crystallization can be attributed to low fiber-matrix interface strength which will prevent matrix cracks from propagating into the reinforcement fibers. However, the maintenance of a low fibermatrix strength causes a low composite interlaminar shear strength.
The glass matrix system has the ability to redistribute loads more effectively at higher temperatures than conventional organic matrix composites. At high temperatures the glass/glass composite will deform and not fracture.
High levels of performance in the interfacial region between the fiber and matrix can be artificially created. Experiments have been conducted using an aluminum metal coating on Si02 fibers which is incorporated in a low temperature glass matrix. The composite produced was a notch insensitive impact resistant material.
These composites must be treated as three component systems, i.e. fiber, matrix and fibermatrix interfacial region. It is this last region which appears to control the fracture process in these composites and hence their relative toughness. A strong fiber-matrix interface permits the early cracks, formed in the matrix, to propagate straight through the entire composite.
If the fiber-matrix interfacial strength is weak enough to prevent cracks in the matrix from propagating into the fiber, however, it is possible to achieve significantly stronger composites. Thus matrix cracking can occur throughout the composite without causing immediate failure.
Since lunar raw materials are available for producing reinforcement glass fibers, it seemed likely that a glass or ceramic matrix would require less basic development. Using glaze and enamel technology as a starting point, the availability of elements that would reduce the melting point of the basic glass formulation was explored.
Glasses used in the manufacture of reinforcing fibers have a working temperature of about 1000 C; the effort here is to explore the formulation of frit-like materials having a low viscosity below 500 C. If this objective proves feasible, the result would be a composite that would be processed in much the same fashion as the very high melt advanced polymer composites currently being used in the airframe/aerospace industry.
Glazes are thin, generally homogeneous glass mixtures fused on the surface of another material. The materials used in the mixing of glaze batches are classified chemically as:
1. BASES, the fluxing agents, which are of the form represented by the RO and the R2O classification and include the alkalies, the alkaline earths, zinc oxide and lead oxide. The alkalies used in glazes are compounds of sodium, potassium and lithium. The alkalies are strong fluxes. They increase the fluidity of the molten glazes.
2. INTERMEDIATES, which include amphoteric oxides of the R2O3 classification, a group which alumina is the common example and to which ferric, chromic manganic, and other oxides are sometimes assigned.
3. ACIDS, which are represented by the R02 classifications, of which silica is the primary type and to which phosphoric oxides, zirconia, and fluorine also belong.
The general molecular equivalent formula for low melting temperature glasses is:
RO + R2O3 + R02
A specific molecular equivalent formula for a frit-like lead glass which melts at 585 C is:
PbO + 0.1 Al203 + 1.5 Si02
Low temperature glasses used on Earth depend on relatively large amounts of lead oxide and boron oxide to reduce the working temperature of the resultant glasses. No boron oxide has been detected on the lunar surface and only small amounts of lead oxide have been observed. Lead oxide does occur in local deposits of vitreous materials as a thin coating of volatile-rich materials (sulfur, chlorine, zinc and lead) on ordinary mare basalts. While many of the fluxing compounds used on Earth were detected in the Apollo samples; no large concentrations of those materials were uncovered at the landing sites. Exploration will be necessary to find deposits of materials which can be refined in order to combine with feldspar to produce very low temperature melting matrix glasses.
The resultant low melting glass formulations used in the environment of space may be very different and less complex than those used on Earth because in many ways the performance requirements are less severe. Several constraints which influence the glass formula for typical Earth products and are not as important in the space environment are the following:
1. The ability to transmit light and other radiations
2. Electrical resistance
3. Forming characteristics for molding
4. Chemical durability-resistance to weathering and chemical attack
5. Color
6. Texture
From a composite fabrication point of view the glass matrix composites, as compared to other ceramic candidates, probably offer the greatest commercial potential. This is true because of their ease of densification, low cost and the achievement of high performance properties. The following attributes are important in this sense:
• Glasses can be created with a broad range of compositions to control fiber-matrix chemical interaction.
• Glasses can be created with a wide range of thermal expansion coefficients to tailor them to nearly match those of reinforcing fibers.
• The ability to control the viscosity of glasses and to deform them easily under pressure permits the physical densification of fiber reinforced composites without mechanical damage to the fibers.
• The composite densification process can be rapid since glass matrix flow is all that is required.
• Since glass can be treated as a thermoplastic material many of the processes developed for fiber reinforced glasses emulate those previously used for polymer matrix systems.
Another important aspect of the development work with glasses on Earth is that the composites are fabricated in a manner completely analogous to that used for resin matrix composites. The most common process is begun by making monolayer tapes which are cut up to make plys which are then stacked and densified to form the final composite in hot pressing operation. This densification occurs while the glass matrix viscosity is low enough to permit permeation of the interstices between the individual fibers. Because pressure is involved, the densification can take place at lower temperatures than required for complete melting of the matrix glass.
It appears that the conversion of glass/glass prepreg to end product may be simpler than in thermoplastic polymer/glass systems. Although both require simple meltchill processing, the glass matrix should not be subject to the sublimation, oxidation degradation, and chain breakdown problems encountered in polymer heat cycles.
As important as the ability to produce the fiber and the matrix is the method of combining them into a convenient form prior to their conversion into the final product. The glass composite could be processed by either taking the basic fiber through a molten frit bath prior to forming into finished component, or the frit could be deposited in powdered form on the fiber with the proper volume fraction and sintered into a prepreg prior to introduction into the conversion process. The latter approach should be more controllable, more universal, and have less potential for degrading the reinforcing fiber. The technique that will ultimately be used will be developed after studies of the manufacturing technology for the frit matrix show the simplest producible form.
ENERGY SOURCES
Energy sources and processing techniques for producing glass fibers have been the subject of previous studies. It is believed that simpler process technology can be developed, particularly in light of investigations to see if composite material production could be tied to the oxygen production process.
It would be desirable if all of the energy for processing both the fiber and matrix materials could be obtained from solar concentrators, and this may be possible. However, it might be worthwhile to use a combination of solar concentrators and electricity in order to better control the temperature of the platinum bushings used for fiber production.
CONTAINERLESS PRODUCTION
In the manufacture of glass fiber on Earth, the factor that causes the most manufacturing difficulties is the solution of the refractory lining the melt tanks into the glass melt itself. These impurities are the primary cause of fiber breakout, as well as a number of other production problems. An intriguing possibility, both on the lunar surface and in space, is the potential for eliminating these problems and achieving super-pure glass by eliminating the need for the refractory container. It is conceivable that in space the melt could be produced without the need for any constraining vessel. This could lead to production rates and fiber properties not possible on Earth. Even on the lunar surface there is the possibility that the basic melt could be used as the container, thereby eliminating contamination of the production melt.
While it is true that the production of glasses in space or in the lunar environment will present many challenges, it is also probable that many of the problems faced in producing high quality glasses on Earth will not be present in the vacuum of space. The primary purpose of the conventional large containers for glass melting is to allow sufficient time for the floatation of bubbles to the surface of the melt. The removal of residual gas bubbles takes much longer than the time for the glass to become completely molten. On Earth the glass making raw materials are comprised of compounds containing gasses (such as carbonates, sulfates, hydrates) and minerals with absorbed gasses. Large volumes of gasses must be removed from these melts. This would not be a problem in space since the lunar materials contain only small amounts of gas and the high vacuum would aid the de-gassing.
CONCLUSION
To date almost all studies have centered on the mining and refinement of traditional metallic structural materials processed from the lunar regolith. This investigation has concluded from a detailed review of the composition of the regolith samples brought back on the various Apollo missions, that the raw material required to produce both the reinforcement fibers and the matrix materials of glass/glass composites are available. Since neither matrix nor reinforcing fibers required a high degree of refinement compared to metals, and both can be processed in extremely small facilities, the negative economics that apply to metals do not apply to composites. Therefore glass/glass composites have tremendous economic and technical advantages over the traditional metallic materials. This investigation concludes that composites may represent a better alternative for producing most of the materials of space construction, both for the establishment of a lunar colony and export to other construction sites in space.
Meeting Notice Midwest Space Development Conference
The second annual Midwest Space Development Conference (MSDC) is scheduled for October 17-19, 1986, at the Strongsville, Ohio Holiday Inn. The hotel is five minutes from the Cleveland Hopkins Airport and NASA Lewis. MSDC will feature a special programming track for educators. Speakers include: Ohio Teacher-in-Space finalist, Gail Klink; Dr. Lynn Bondurant of NASA; and Phillip Woodruff of the Young Astronaut Program. The parallel technical track will include presentations by Gregg Maryniak on SSI’s research and reports from NASA Lewis.
On Saturday, October 18 from 9 to 11 pm you are invited to the SSI hospitality room, cosponsored by the Ohio, Wisconsin, Chicago, Pittsburgh and Minnesota Support Teams.
For more information please write: MSDC, 2720 West 40th Street, Lorain, Ohio 44053
(xxx-xxx-xxxx).
©space studies institute