SPACE STUDIES INSTITUTE
P.O. BOX 82
PRINCETON, NEW JERSEY 08540
[[librarian note: This address is here, as it was in the original printed newsletter, for historical reasons. It is no longer the physical address of SSI. For contributions, please see this page]]
SSI UPDATE
THE HIGH FRONTIER® NEWSLETTER
VOLUME XII ISSUE 4 JULY/AUGUST 1986
VICE PRESIDENT’S COLUMN
In all of our communications, SSI invites you to participate in the human breakout into space through your support of our ongoing research. In this newsletter we have two very special reports on projects made possible by your efforts.
In June, SSI’s research on Solar Power Satellites (SPS) constructed from lunar materials was presented at Paris, France. It is interesting to note that interest in SPS is now especially strong in Europe, Japan and the Soviet Union, with the Soviets planning some sort of SPS by the year 2000. SSI’s work showing the dramatic reduction in SPS cost made possible by lunar materials is particularly important today. Scientists around the world are deeply concerned about the effects of fossil fuel combustion on the Earth’s biosphere. As the demands for energy increase, particularly in the third world, SPS may provide a key option for preservation of our planet.
The long-awaited National Commission on Space Report has been made public and it contains important elements of all of SSI’s critical-path research. This report constitutes a “Seal of Approval” for the ideas that we have all worked so hard to support and demonstrate. Each one of us can look back at our individual contribution to the SSI effort and see it reflected in this important document. Thank you for making this research possible! Sit back and enjoy reading the results of your participation in the Institute.
Gregg Maryniak
Space Studies Institute Relocates
Effective August 1st Space Studies Institute will be located in the Princeton Business Park in Rocky Hill, NJ. Rocky Hill is a small community just north of Princeton. We will continue using our mailing address of P .0. Box 82, Princeton, NJ 08540 and phone number, xxx-xxx-xxxx. If you plan to visit the Institute, please call for directions.
SSI BOARD MEMBER TO LEAD NASA
Dr. James C. Fletcher, appointed by President Reagan to head NASA, is not only a former NASA administrator but is also a former member of the Board of Trustees of the Space Studies Institute. Dr. Fletcher served on our Board until his appointment to his new post. We appreciate his support and guidance and wish him the best of luck with his work at NASA.
RESEARCH REPORT
COMPOSITES: Fibers and Matrices from Lunar Regolith
By Charles H. Coggin, Jr.
President of Glass Strand, Inc.
Mr. Coggin is principle researcher for SSI’s composite research project. The following is an excerpt from the interim report submited by Glass Strand, Inc. Part two will be featured in the September/October issue of Update.
Work now underway on space stations almost certainly portends an era of ever increasing construction in space. The Moon could become economically self-supporting by supplying raw materials for the construction and operation of lunar colonies and space stations. It has been proposed that lunar rocks and soils will be useful raw materials for manufacturing products for use in space construction both on and off the lunar surface. The lunar regolith (soil) will be strip mined. Front end loaders would dig out the soil and deliver it to an automated plant where different minerals would be separated. The benefication of ores to an industrial feedstock grade is always desirable before such material is chemically processed, both because it reduces the size and energy requirements of chemical processes and because it can significantly reduce the complexity of such processes while increasing their efficiency.
More than a ton of moon rocks brought to Earth by astronauts proved that the Moon is a rich source of useful materials. However, nobody is talking about mining resources on the Moon and shipping them back to Earth. But scientists are looking at lunar materials for use at Moon bases and for export to space stations. One of the most valuable lunar materials is oxygen. Lunar rocks are roughly half oxygen. It is not free oxygen, but chemically combined with a variety of other elements including titanium, calcium, aluminum, iron, silicon and magnesium. Oxygen means more than just life support in space. The most efficient rockets are fueled by hydrogen and oxygen. Liquid oxygen will be a major item of commerce in space and liquid oxygen production will be the chief industry of the Moon base. Calculations indicate that the energy needed to lift a pound of material from the Lunar surface into space is less than one twentieth that of the cost of lifting a pound from the Earth’s surface into space.
Fiber-composites can also be produced from materials available from lunar soil directly and from by-products of oxygen, aluminum and titanium extractions. Fiberglass is an ideal material for use in space construction: it can be used for manufacturing many lunar and space station structural components such as reinforced structural panels, large fiberglass cylinders, and small-diameter pultruded tubing for struts.
A fiber-composite material is one consisting of two or more discrete physical phases, in which a fibrous phase is dispersed within a continuous matrix phase. The fibrous phase must retain its physical identity, such that it conceivably can be removed from the matrix intact. Fiber-composite materials are both complex and versatile. They are complex by virtue of their chemical and mechanical nature and thus should be regarded not as a single material, but as a materials system. Their versatility stems from a wide choice of constituent materials available and from the variety of ways in which composites can be fabricated to provide a combination of desired properties that cannot be achieved in conventional materials.
The technology of high performance fiber-composites is closely related to that developed in glass-reinforced plastics. However, the necessity to bring organic plastic feedstocks from Earth to provide the matrix material is not as cost effective as using matrix material from the Moon.
The purpose of this study is to determine if lunar surface materials are present to provide feedstocks for:
1. High temperature glass fibers
2. Low melting temperature glass to be used as a matrix for fiber-composites
This study will develop conclusions based on the analysis of Apollo and LUNA regolith samples returned to Earth as compared to the necessary ingredients to form glass fibers as produced on Earth.
LUNAR RAW MATERIALS
The surface of the Moon consists of two major physiographic units: the higher albedo (brighter) highlands and the lower albedo (darker) maria.
HIGHLANDS The lunar highlands that have been sampled consist primarily of feldspar-rich rocks that have been intensely brecciated and locally melted by intense impact cratering.
MARIA The maria were formed by filling of the mare basins and other low areas by basaltic magma. The rocks thus formed are a series of iron and titanium rich basalts. The mare basalts are enriched in some elements (barium, strontium, rare earth elements, uranium) but generally are depleted in relatively volatile elements, in particular potassium, sodium and lead.
LUNAR SOIL The surface of the maria is covered to a depth of 5 to 15 meters by a fragmented soil (the regolith) produced by repeated impacts. The highlands, more heavily cratered, have a much deeper regolith. Most of the mare regolith is locally derived, but it contains a minor component of exotic fragments ballistically transported by impacts from distant areas.
If non-terrestrial materials are to be used for glass fiber composite materials, it is of considerable importance to characterize their physical and chemical properties.
Lunar materials may be further classified as follows:
(1) Regolith, a fine-grain deposit loosely referred to as “lunar soil”
(2) Igneous rocks that were derived from the Moon’s interior by well-known igneous processes
(3) Breccias, which represent lunar deposits that were lithified by the effects of meteorite impact
Because of the numerous impacts in the regolith; it is highly comminuted and very rich in glass. Lunar soils consist mostly of (1) lithic and mineral debris derived by impact comminution of the underlying bedrock and (2) glass particles formed by impact melting.
The dominant minerals on the lunar surface are pyroxenes, olivines, plagioclase feldspars, ilmenite and spinets. The pyroxenes, olivines and plagioclase are silicates. Chemical analysis of typical lunar materials list Si02 contents of approximately 50% for pyroxenes, 37% for olivine and 46% for plagioclase. Pyroxenes are being considered as possible sources of magnesium, calcium, iron and aluminum; olivines are a potential source of magnesium and iron; and plagioclase is a potential source of aluminum.
It is important to note that many fundamental questions about the Moon are yet to be answered. Only about half the major types of basalts are represented in Apollo and Luna samples. Man has yet to visit or sample the far side. The highlands are virtually unexplored. Because of Apollo latitude restrictions, the polar regions have not been visited. A variety of unsampled surface features that may have importance in resource considerations warrants investigation, such as the dark halo craters (possible volcanoes with volatile material and/or deep-seated fragments).
The mare regions cover 17 percent of the Moon’s surface and consists primarily of basaltic volcanic rock that has spread as relatively thin, widespread flow units. Local deposits of vitreous materials that apparently are equivalent to terrestrial volcanic ash have been found. Whereas these materials have the bulk chemical composition of basalt, they also oontain thin coatings of volatile-rich (sulfur, chlorine, zinc and lead) materials, strongly enriched with respect to ordinary basalts.
The composition of the non-mare regions is complex and highly variable. The predominant crystalline rock types are from a suite of materials that have been termed the anorthosite-norite-troctolite suite. The principal rock forming minerals are similar to those of the mare basalts. Amorthosites are rich in the mineral plagioclase; norites are plagioclasepyroxene rocks; and troctolites are plagioclaseolivine rocks. In detail the mineral compositions are distinct from the mare basalts in that the pyroxenes and olivines have the lower iron/magnesium ratios and the plagioclase contains less sodium. The oxide minerals such as ilmenite are rare.
Several classes of possible primary rock types were observed in the lunar samples and have been inferred to exist in abundance on the basis of Apollo orbital data. These include the variety known as KREEP, enriched in Potassium, Rare Earth Elements and Phosphorus. They are of importance because many trace elements are enriched in these relative to typical lunar basalt compositions.
The chemical composition of the regolith varies with grain size. The less than 10 micron fraction is chemically fractionated from the bulk soil. In most cases the less than 10 micron fraction is enriched in M2O3, CaO, Na2O, K2O and light rare earth elements. This is largely a result of simple comminution, with feldspar from local source rock concentrating in the finest fraction. There is evidence that agglutinate glass forms preferentially by fusion of the finest soil fractions.
A comprehensive review of the chemical analysis data of samples returned to Earth from the six Apollo and three Luna missions was conducted during this study in order to determine the availability of both high temperature glass fiber raw materials and low temperature glass matrix materials. Apollo 11 and 12 and Luna 16 and 24 were mare sites; Apollo 15 and 17 were combined mare and highland sites. Apollo 16 was the best highland site, located in the central highlands.
GLASS FIBERIZATION REQUIREMENTS
The number of possible glass formulations is almost infinite. However, a definite limitation apparently does exist; since it would appear that in all glass compositions, at least one of a very small class of substances must be present. Thus, although almost any element in the periodic table could be present in a glass, very few compositions would be found that did not contain silicon, boron or phosphorus in considerable quantity. Silicon in its oxide form is the only glass forming element found on the lunar surface. Other oxides which approach glass forming oxides, but which do not quite qualify, are called intermediates and those which are practically devoid of the glass forming tendency are called modifiers.
Another scheme of classification of the common oxides considers them in the three following groups:
(1) glass formers
(2) stabilizers
(3) fluxes
Fluxes are those oxides which react in the glass batch at relatively low temperature, generally Group I alkaline oxides. Glasses high in such fluxes tend to have low chemical resistance. Stabilizers are those oxides which impact to the glass a high degree of chemical resistance. While an almost infinite number of glass compositions are possible only a much smaller number of compositions will form glasses which can be fiberized.
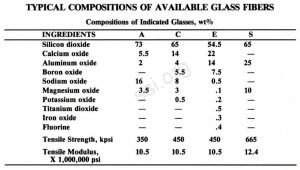
There are four general commercial types of glass fibers produced on Earth. These are known as A, C, E, and S glass. Only A, C, and E are produced in large quantities.
There is presently in use a replacement glass for “E” which doesn’t require Boron Oxide as a flux. Boron and fluorine have been recognized as potential pollutants in the manufacture of fiberizable glasses and have been eliminated by substituting Ti02, MgO and ZnO in the basic three component glass composition of SiO2, AlSO3 and CaO.
One primary limitation effecting the melting and fiberization of potential compositions is the maximum use temperature of the melting and fiberizing equipment. Fiberizing methods require that glass must display a relatively low temperature coefficient of viscosity at a viscosity of approximately 1000 poises. A further disadvantage is that the fiberizing temperature is determined by the viscosity characteristics of the glass. Also, the surface tension of the melt at the fiberization temperature must be lower than the viscous drag; otherwise the glass cannot be extended into a fiber, but will separate into distinct droplets.
Current methods to fiberize glass impose certain stringent property relationships on the glass compositions. Particularly important are the temperature-viscosity, surface tension, and crystallization rate relationships.
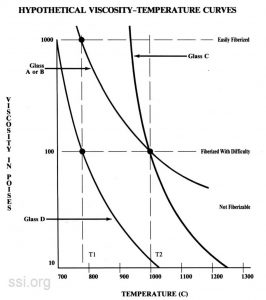
The temperature-viscosity curve and the liquidus temperature relationship for Glass A represents conditions for easy fiberization. The viscosity at the liquid us temperature is 1000 poises, which is sufficiently high to inhibit rapid crystal growth and is in the favorable range for fiber drawing. Also, the temperature coefficient of viscosity at liquidus temperature is sufficiently high so that slight cooling below liquidus temperature results in a rapid increase in the viscosity which further inhibits crystallization. The ideal glass has a temperatureviscosity relationship possessing a rapid change in viscosity with a slight change in temperature preventing crystal growth in the fiber state.
Glass B is represented as having the same viscosity-temperature curve as Glass A, but liquidus temperature is located at a temperature representing 100 poises. This glass would be difficult to fiberize; a viscosity of 100 poises allows rapid crystallization. The temperature coefficient of viscosity at liquidus temperature is low, so that an extremely sharp temperature gradient temperature (liquidus temperature = 1000 poises) would be required to prevent divitrification.
Glass C would be easier to fiberize than Glass B, even though their liquidus temperatures are identical and their viscosities at liquidus temperature are the same. This is because the temperature coefficient of viscosity of Glass C is higher than that of Glass B at liquidus temperature. Glass C would be more difficult to fiberize than Glass A because of its low viscosity of the liquidus temperature.
Glass D represents the same condition as Glass c, except that its liquidus temperature is lower, and is represented as being the same as for Glass A. Glass D would be more difficult to fiberize than Glass A because of its low viscosity at liquidus temperature; easier to fiberize than Glass B because of its higher coefficient of viscosity at liquidus temperature; possibly less expensive to fiberize than Glass C because the lower furnace temperature represents a longer bushing life.
The second half of this report will be presented in the September/October Update.
SSI WELCOMES NEW CORPORATE MEMBERS
The Institute is pleased to welcome two new corporate members in this issue. Friesema & Associates, Inc. is an independent microcomputer consulting firm based in Oak Park, Illinois. Bill Friesema, the firm’s president, was recently featured in a New York Times article on professionals who work from their homes. Friesema & Associates, Inc. has clients throughout the United States. Bill and his company have been longtime supporters of SSI and we are proud to include this firm in our family of corporate supporters. For more information on his organization call xxx-xxx-xxxx.
SSI also welcomes another close friend into our corporate membership program. Space Age Publishing Company of Santa Clara, California, best known for its weekly publication, Space Calendar, has become our newest corporate member. Publisher Steve Durst is well known within the space movement for his rapid reporting of events of interest within our community. Space Calendar is now offering a special introductory rate for its publication for SSI supporters. For more information call xxx-xxx-xxxx.
SSI REPLICATORS ANNOUNCED
We are proud to announce the following people have been added to the list of SSI Replicators:
Bill Weigel
David Wine
Mike Massimino
Dave Brody
These Senior Associates have signed up either one new Senior Associate or Corporate Member or five new subscribers. Congratulations and thanks to each new SSI Replicator.
NEW SUPPORT TEAMS FORMED
Philadelphia, Pennsylvania and Madison, Wisconsin are now SSI Support Team locations thanks to the efforts of two Senior Associates. Randy Gigante, an engineer and student at the Wharton Business School, has taken on the task of organizing a team in the City of Brotherly Love. Ann Bennett, a paralegal assistant in Madison, has agreed to head up SSI efforts in that city. Both Ann and Randy would be happy to hear from other SSI supporters and can be reached through the Institute office.
THE NATIONAL COMMISSION ON SPACE REPORT
PIONEERING THE SPACE FRONTIER
Pioneering the Space Frontier is available at a discount through Bantam books when ordering four or more copies. To order send $8.37 per copy in check or money order with your name and UPS shipping address, noting that you are an SSI member, to: Steve Mudgett, Bantam Books, Special Sales Department, 666 5th Avenue, NY, NY 10103.
Delivery will be approximately two weeks after receipt of your order. If you have any questions you may call Bantam at: xxx-xxx-xxxx.
For single copies, you may order through SSI. Send your name, address and check for $11.00 per copy to: SSI, P.O. Box 82 Princeton, NJ 08540
Bettie Greber Named Editor of SSI Update
On behalf of the President and Trustees of the Space Studies Institute, I am pleased to announce that Bettie Greber has been named Editor of SSI Update. This title gives official recognition to the fact that Bettie leads the team effort that goes into editing and producing the Institute’s primary communications tool. Congratulations, Bettie!
Gregg Maryniak
SSI TO COSPONSOR SPACE CONFERENCES IN 1986
The Space Studies Institute is formally cosponsoring two Space Conferences in Fall, 1986. The first, scheduled for September 22-24 in Atlantic City New Jersey is entitled The First Lunar Development Symposium. The prime sponsor of the Conference is the American Maglev Company. Speakers at the Symposium will include Dr. Isaac Asimov, Dr. Henry Kolm, Dr. Michael Duke and Gregg Maryniak. For more information, call xxx-xxx-xxxx, or write:
Symposium ’86
P.O. Box 1986
Pitman, NJ 08071
The second Conference is the annual American Astronautical Society Conference in Boulder, Colorado. The dates for this conference are October 26-29, 1986. Gregg Maryniak will present a paper on SSI’s research and SSI Trustee Martin Rothblatt will chair the session on commercial satellites and present a paper on Geostar. For additional information, call xxx-xxx-xxxx, or write:
Dr. W. Morgenthaler
University of Colorado
Campus Box 423
Boulder, CO 80309
©space studies institute